Manufacturers and operators in the packaging sector, within their research, design, and quality control departments, can now rely on a valuable tool to
identify and assess various parameters and potential defects related to the packaging process, even when implemented in CAP (Controlled Atmosphere
Packaging) or MAP (Modified Atmosphere Packaging) systems.
Thanks to the Prescale film, a thin layer that can be easily inserted and positioned between contact surfaces, it is possible, for example, to verify the
correct alignment of the sealing matrix and ensure that the packaging materials are compressed correctly and, most importantly, uniformly.
This enables qualitative checks characterized by good repeatability.
Several operators in the pharmaceutical and food packaging sectors use Prescale film to ensure, for example, that the pressure from the thermoforming
machine's plate is sufficient to seal the blister. They perform this type of verification periodically as a quality control measure, "recording" the
results in the form of the image impressed on the film, or when a new product or a new thermoforming set is introduced.
In addition to allowing the easy optimization of blister packaging production, this control methodology also helps prevent any unwanted "grooves" or
indentations that could lead to poor sealing (leakage) between the two sealing materials of the blister, and consequently the premature deterioration
of the contents (e.g., capsules, pills, food products, etc.).
In response to these needs, Fuji Film offers a special film that can be "impressed" based on the imprint exerted on it, creating an unmistakable record that allows for the precise identification of contact areas and the pressure
applied to every minute portion of the contact surface.
The Prescale film is essentially composed of a layer of film onto which an extremely homogeneous and uniform layer of microcapsules containing a coloring
substance (PSC - Particle Size Control technology) has been deposited. Additionally, another layer of film is applied, containing a specific material
necessary for the development and stabilization of the colorant. When pressure is applied to the Prescale film, it causes some of these microcapsules
to break. Depending on their number and the compression force exerted, they can color the other section of the film (with varying shades of red, the
density of which is evidently proportional to the pressure applied), which is intended for the development and proper fixing of the colorant.
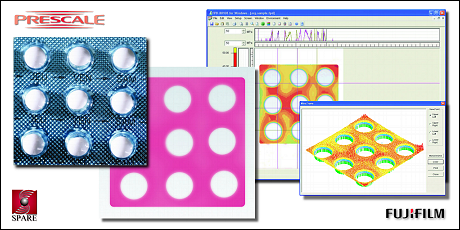
The film can be cut to size with simple scissors and placed in the area of interest for both static and dynamic measurements. By then performing a visual
inspection using colorimetric tables provided with the film (or with suitable electronic analysis tools available separately—see the image below, which
is correlated with false colors), it is possible to identify the different color gradations that have developed on the Prescale film. This allows the
operator to produce a documented test report and also offers the possibility of creating a historical record of inspections conducted over time.
The Prescale film is available in eight different gradations (5LW, 4LW, LLLW, LLW, LW, MW/MS, HS, HHS), which cover measurement ranges from 0.006/0.05
MPa to 130/300 MPa. For all versions, a measurement accuracy of no worse than +/-10% FS is guaranteed (which can be significantly improved when the
film is used in conjunction with appropriate analysis instruments). More details and technical literature can be obtained by visiting the website
www.prescale-film.com.
You are here: press · prescale blister Description: Press Release: Blister Packaging Verification and Analysis
- editorial prepared by: Spare sas
- prepared by (author): ufficio marketing
- section:: Press Releases and Technical Articles
Press Release
Blisters (food & pharma packaging) - Blister Packaging Verification and Analysis
Manufacturers and operators in the packaging sector, within their research, design, and quality control departments, can now rely on a valuable tool to identify and assess various parameters and potential defects related to the packaging process, even when implemented in CAP (Controlled Atmosphere Packaging) or MAP (Modified Atmosphere Packaging) systems.

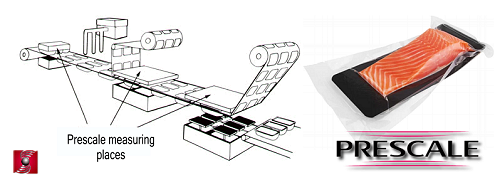
Thanks to the Prescale film, a thin layer that can be easily inserted and positioned between contact surfaces, it is possible, for example, to verify the correct alignment of the sealing matrix and ensure that the packaging materials are compressed correctly and, most importantly, uniformly. This enables qualitative checks characterized by good repeatability.
Several operators in the pharmaceutical and food packaging sectors use Prescale film to ensure, for example, that the pressure from the thermoforming machine's plate is sufficient to seal the blister. They perform this type of verification periodically as a quality control measure, "recording" the results in the form of the image impressed on the film, or when a new product or a new thermoforming set is introduced.
In addition to allowing the easy optimization of blister packaging production, this control methodology also helps prevent any unwanted "grooves" or indentations that could lead to poor sealing (leakage) between the two sealing materials of the blister, and consequently the premature deterioration of the contents (e.g., capsules, pills, food products, etc.).
In response to these needs, Fuji Film offers a special film that can be "impressed" based on the imprint exerted on it, creating an unmistakable record that allows for the precise identification of contact areas and the pressure applied to every minute portion of the contact surface.
The Prescale film is essentially composed of a layer of film onto which an extremely homogeneous and uniform layer of microcapsules containing a coloring substance (PSC - Particle Size Control technology) has been deposited. Additionally, another layer of film is applied, containing a specific material necessary for the development and stabilization of the colorant. When pressure is applied to the Prescale film, it causes some of these microcapsules to break. Depending on their number and the compression force exerted, they can color the other section of the film (with varying shades of red, the density of which is evidently proportional to the pressure applied), which is intended for the development and proper fixing of the colorant.
The film can be cut to size with simple scissors and placed in the area of interest for both static and dynamic measurements. By then performing a visual inspection using colorimetric tables provided with the film (or with suitable electronic analysis tools available separately—see the image below, which is correlated with false colors), it is possible to identify the different color gradations that have developed on the Prescale film. This allows the operator to produce a documented test report and also offers the possibility of creating a historical record of inspections conducted over time.
The Prescale film is available in eight different gradations (5LW, 4LW, LLLW, LLW, LW, MW/MS, HS, HHS), which cover measurement ranges from 0.006/0.05 MPa to 130/300 MPa. For all versions, a measurement accuracy of no worse than +/-10% FS is guaranteed (which can be significantly improved when the film is used in conjunction with appropriate analysis instruments). More details and technical literature can be obtained by visiting the website www.prescale-film.com.
©1998-2024 Spare sas
In preparing this document, the utmost care has been taken to ensure that the content accurately reflects the characteristics of the described product. However, despite these efforts, Spare sas cannot be held responsible or liable for any inaccuracies. Furthermore, in the context of ongoing product development and improvement, please note that the information provided may not be up-to-date with the latest version, as manufacturers reserve the right to withdraw or modify their product documentation at any time without notice. Consequently, it is advised to verify the accuracy and currency of the information at the time of purchase.